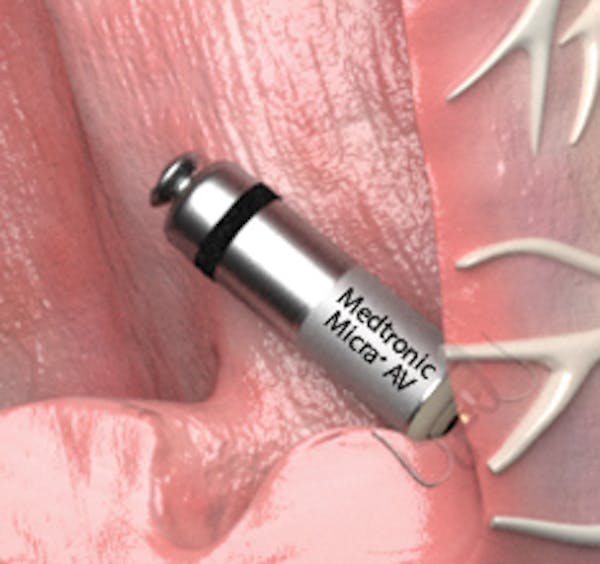
Leadless Pacemaker “Micra” Implantation - Phoenix, AZ & Tempe, AZ: Arizona Heart Arrhythmia Associates
India, too, red-flags 3 pacemakers made by US firm Medtronic for sudden ' battery depletion' - The Hindu BusinessLine
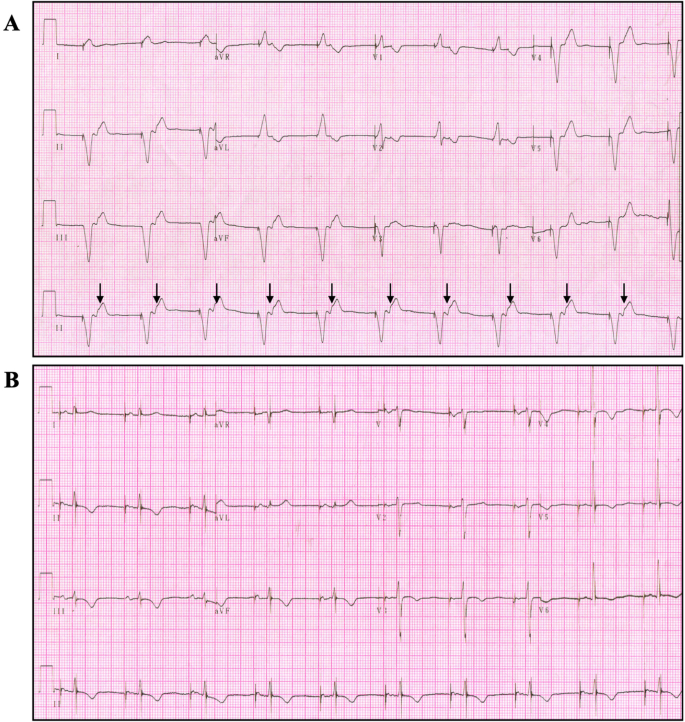
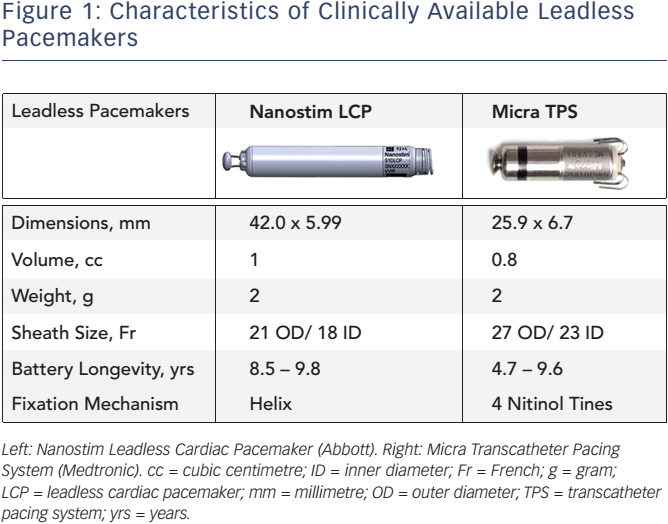
Adverse clinical events caused by pacemaker battery depletion: two case reports | BMC Cardiovascular Disorders | Full Text

FDA Alert Indicates Some Medtronic Pacemaker Batteries Can Fail Without Warning | MedTruth - Prescription Drug & Medical Device Safety | Informed Advocacy







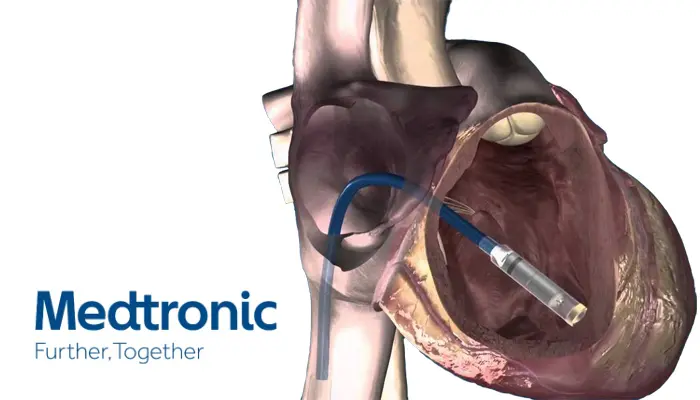
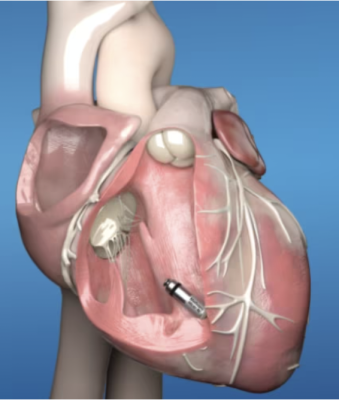
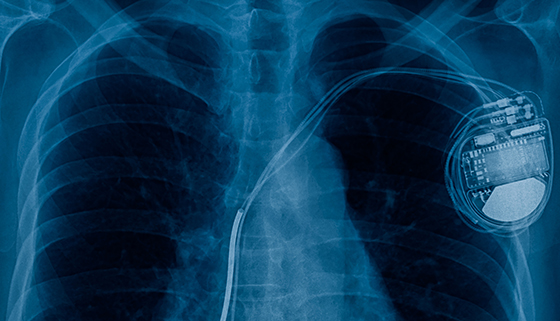
/arc-anglerfish-arc2-prod-dmn.s3.amazonaws.com/public/AT25LTER4G66PEKAEFOT53DQGI.png)