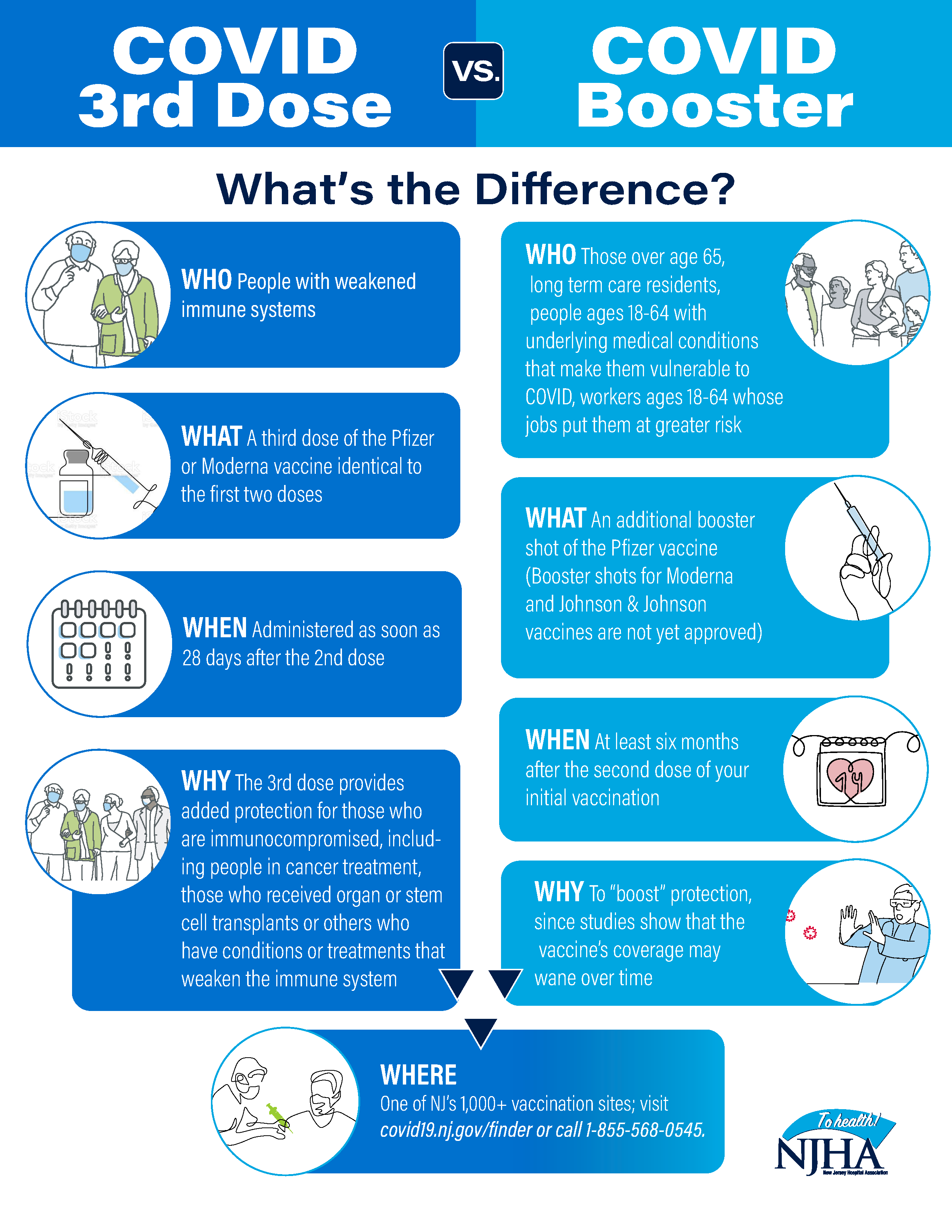
COVID-19 Vaccine Boosters vs. Third Doses: Frequently Asked Questions - Anne Arundel County Department of Health

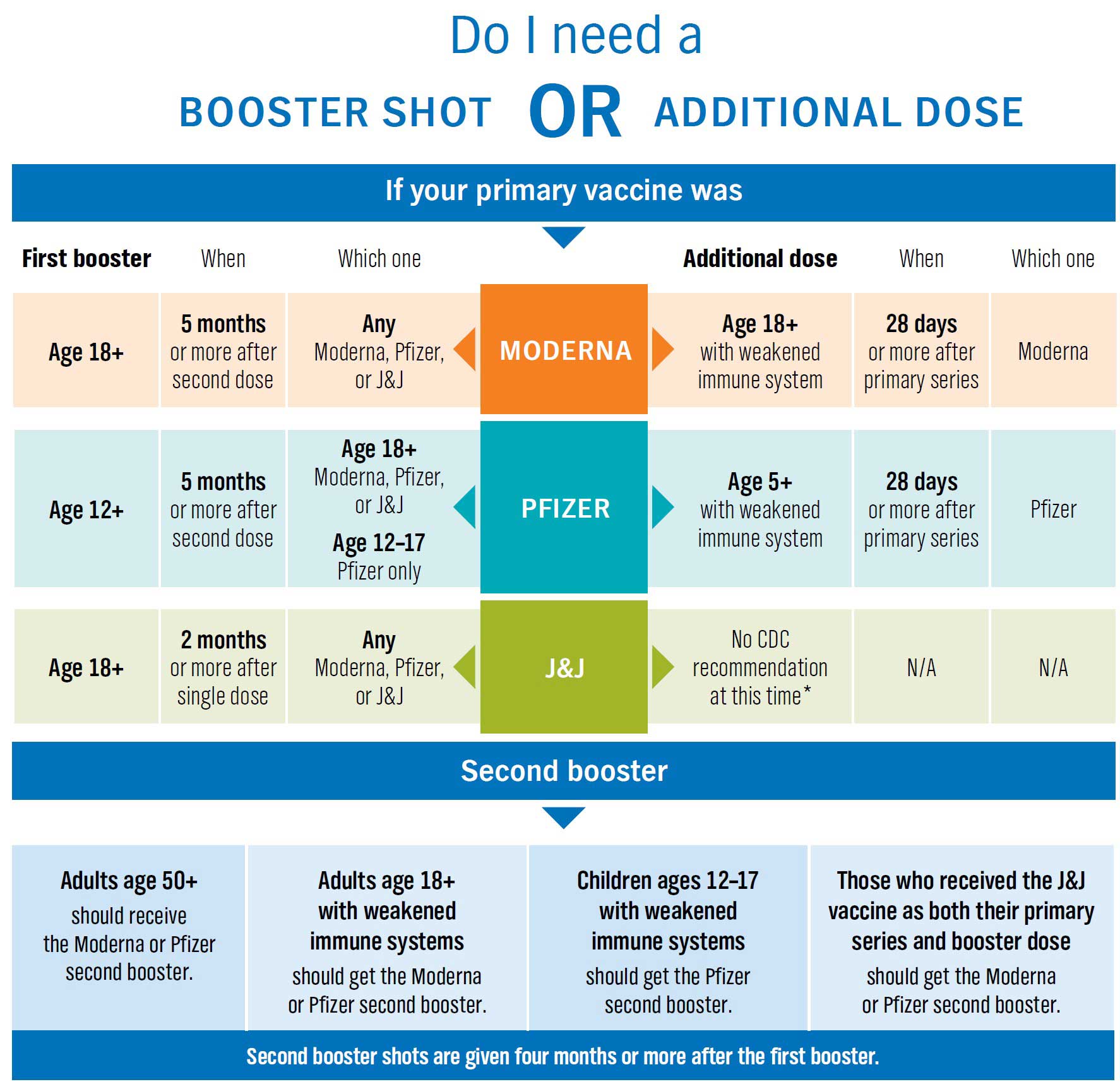
Coronavirus (COVID-19) Update: FDA Shortens Interval for Booster Dose of Moderna COVID-19 Vaccine to Five Months | FDA

DeKalb County Health Department - What you need to know about an Additional COVID-19 Dose vs. Booster Dose. | Facebook

Early Estimates of Bivalent mRNA Booster Dose Vaccine Effectiveness in Preventing Symptomatic SARS-CoV-2 Infection Attributable to Omicron BA.5– and XBB/XBB.1.5–Related Sublineages Among Immunocompetent Adults — Increasing Community Access to Testing ...

Immunogenicity and safety in healthy adults of full dose versus half doses of COVID-19 vaccine (ChAdOx1-S or BNT162b2) or full-dose CoronaVac administered as a booster dose after priming with CoronaVac: a randomised,
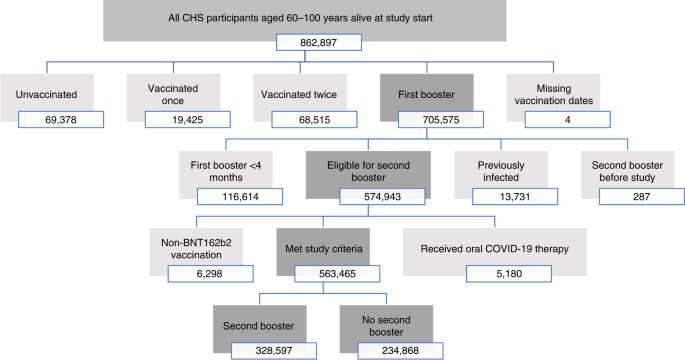



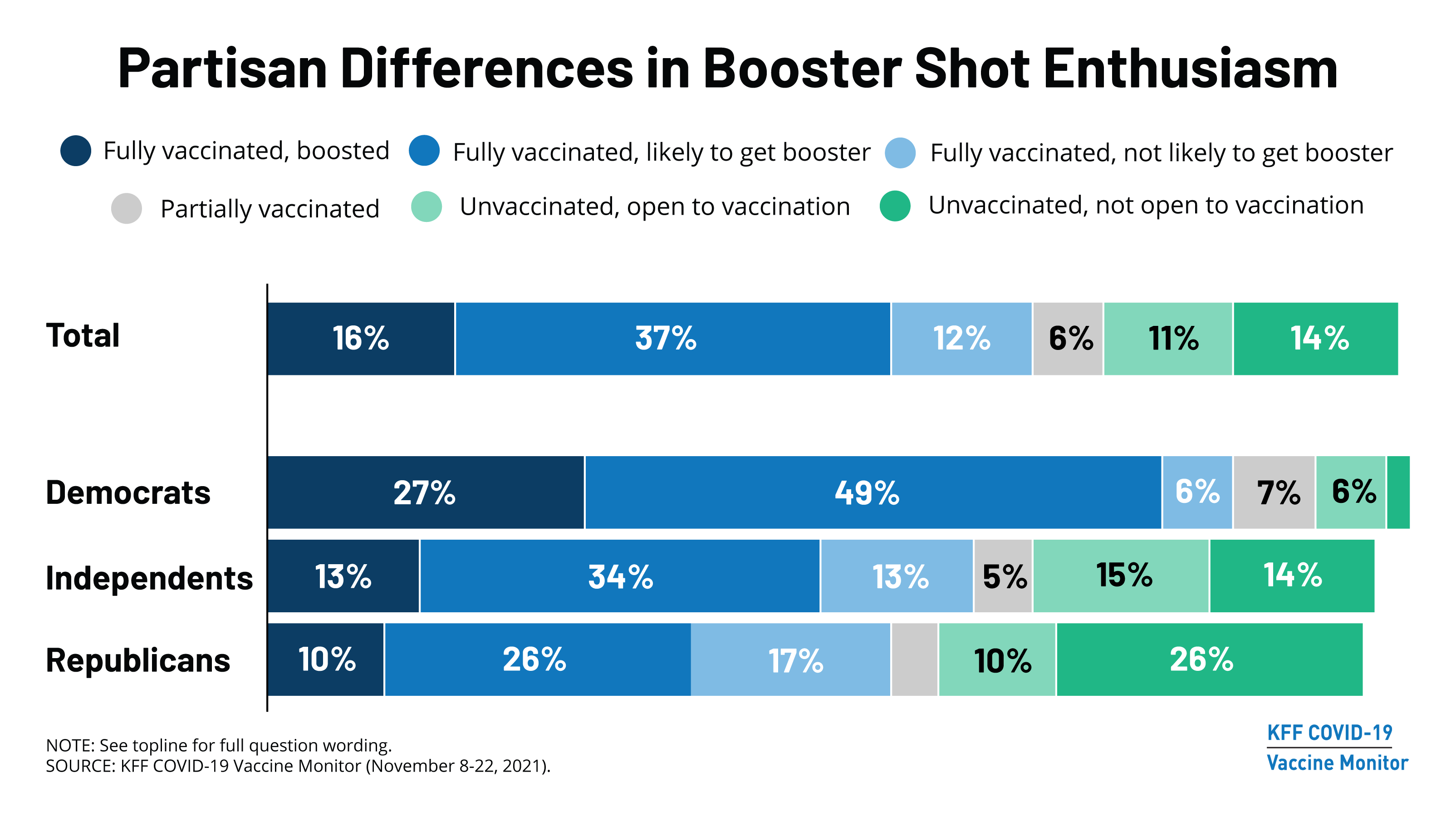


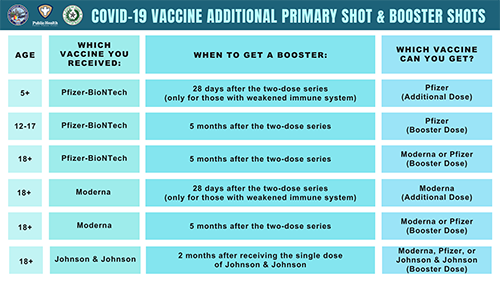


/cloudfront-us-east-1.images.arcpublishing.com/gray/J75DOEBX45DOJI6AN7AWF3R3SM.png)